Uganda
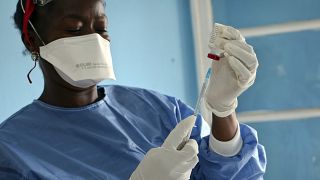
A shipment of Ebola vaccine candidates to be used in a clinical trial has arrived in Uganda.
Health minister Jane Ruth Aceng and the WHO Uganda incident manager attended a ceremony to receive the doses, Thursday (Dec. 08).
Even though the outbreak is seemingly waning with no new cases reported recently, Aceng remained cautious. Indeed, according to WHO criteria, an Ebola outbreak ends when there are no new cases for 42 consecutive days.
"We are nine days today countdown," she explained.
"So it does not mean we will not get another (Ebola) case. Uganda is a country that always wants to be prepared and ready whenever any outbreak occurs."
There are currently no licensed vaccines for the Sudan strain of the virus.
The first batch of one of the 3 candidate vaccines will therefore be evaluated during the Tokomeza Ebola clinical trial.
"The 1,200 doses that the country is receiving today are being or have been manufactured by the Sabin vaccine Institute in the United States," Charles Njuguna said.
The WHO incident manager and doctor of epidemiology added: "They will be administered to those at the greatest risk and what we mean here is the immediate contacts of a person who has been diagnosed with the Sudan virus disease."
The other candidates are a vaccine developed by Oxford University and the Jenner Institute in Britain, the last one came through the International AIDS Vaccine Initiative (IAVI), WHO said.
Since the outbreak was declared on September 20, 56 people died.
According to statistics compiled by Uganda's Ministry of Health, 142 cases were confirmed.
86 people recovered from the hemorrhagic fever. Symptoms include fever, vomiting, diarrhoea, muscle pain, and, at times, internal and external bleeding.
Experience from the far more frequent outbreaks of the Ebola Zaire strain -- for which a vaccine was developed after the massive West Africa outbreak that started in 2013 -- shows that "you can get control much quicker using effective vaccines," WHO emergencies director Michael Ryan told reporters.











01:13
Uganda reopens border with M23-held eastern DRC
01:49
BRICS summit ends on health issues and role of global south countries
01:00
Pix of the Day: July 3, 2025
01:04
South Africa reports new bird flu outbreak on poultry farms
00:50
Ons Jabeur retires from Wimbledon Opener due to breathing issues
01:45
From Uganda to NYC: Zohran Mamdani's rise in American politics