Covid Vaccine
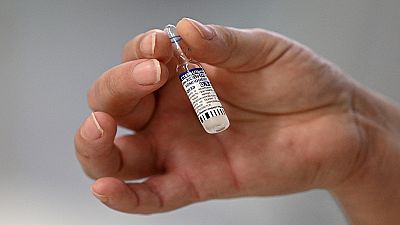
The Africa Vaccine Acquisition Task Team (AVATT), has secured over 300 million doses of Russia’s Sputnik V vaccine.
The AVATT team had earlier on secured over 270 million doses of AstraZeneca, Pfizer, and Johnson & Johnson vaccines in the first allocation phase.
A total of 26 African countries have already submitted their applications to acquire 180 million doses of these vaccines according to AVATT director John Nkengasong.
The AVATT team was set up by the African Union and aims at immunizing 60% of the continental population.
The Sputnik vaccine is 91.6 percent effect against symptoms of Covid-19, according to a report published by the Lancet.
The vaccine was approved in Russia a few months ago from its final-stage clinical trials leading to skepticism from experts.
Several countries around the world have ordered the Sputnik V, according to the Russian investment Fund which helped develop the vaccine.
In January, Chancellor Angela Merkel said Germany had offered Russia support in Moscow's development of Sputnik V after Russian authorities said they had applied for registration in the European Union.
- 'Global response' -
The trial involved giving 14,964 participants in the vaccine group and 4,902 in the placebo group two jabs 21 days apart.
Those taking part were tested for Covid-19 at enrolment into the trial, again when they had the second dose and then after that if they reported symptoms.
From the second dose, 16 cases of symptomatic Covid-19 were confirmed in the vaccine group and 62 cases were reported in the placebo group, giving an efficacy equivalent to 91.6 percent.
The authors said, however, that efficacy was only calculated on symptomatic cases and said more research would be needed to assess how it affects asymptomatic disease.
They added that the follow-up period was around 48 days from the first dose, so the full period of protection is still unknown. The trial is ongoing and plans to recruit a total of 40,000 people.
Sputnik V uses two different disarmed strains of the adenovirus, a virus that causes the common cold, as vectors to deliver the vaccine dose.
Developers said that using a different adenovirus vector for the booster vaccination minimizes the risk of the immune system developing resistance to the initial vector, so it may help create a more powerful response.










01:19
China-Africa entrepreneurs summit calls for deepened cooperation
00:31
Senegal's ex-president Macky Sall urges peace and diplomacy at UN audition
01:12
Togo to ask UN members to adopt world maps that show true size of Africa
01:09
UN slave trade resolution welcomed in West Africa, African Union
Go to video
African Union adopts resolution calling slavery and colonialism genocide
01:01
African Union holds summit: is it ignoring the real issues?